A proposal for the ICD-10 G93.3 legacy terms for ICD-11: Part One
April 2, 2017
Post #329 Shortlink: http://wp.me/pKrrB-4dV
Our Proposal and Rationale is set out in Part Two:
A proposal for the ICD-10 G93.3 legacy terms for ICD-11: Part Two
A version of ICD-11 in 2018
It’s been a long time coming and it ain’t finished yet…
The World Health Organization (WHO) has been revising ICD-10 since 2007.
After several shifts in the timeline, WHO plans to present a version of the next edition (ICD-11 MMS) at the World Health Assembly (WHA), in May 2018.
WHO won’t be seeking endorsement of the ICD-11 product in May 2018 because it won’t be ready to implement. Endorsement will be sought at some point in the future. In the meantime, a version of ICD-11 is scheduled for release later in 2018, after the May assembly. The release date has yet to be announced.
https://hscic.kahootz.com/connect.ti/t_c_home/view?objectId=297939
“…The World Health Organization (WHO) is currently developing the 11th revision of ICD. Once endorsed by the World Health Assembly (WHA), WHO Nomenclature regulations stipulate that Member States must use the most current revision for mortality and morbidity purposes. For this reason and to allow member countries to adopt the new revision when they are ready, WHO will brief the WHA on ICD-11 in May 2018 but will not seek endorsement at this time.”
Member states will transition from ICD-10 to the new edition at their own pace. It’s going to be several years before countries have evaluated the ICD-11 product for utility and prepared their health systems to make the transition.
At some point, data using codes from the new edition will be accepted alongside data compiled using ICD-10. WHO will continue to support ICD-10 until the majority of member states have adopted and implemented the new edition.
It will take even longer for countries like the U.S. and Canada, who use a country specific adaptation of ICD, to implement as they will need to modify the new edition to suit their countries’ health systems. The earliest Canada can implement is currently projected as 2023 [1]. The U.S.’s CDC estimate it will take at least 6 years after the codes have been ratified to prepare, field test and implement an ICD-11-CM/PCS.
Proposal deadlines
Some important deadlines for proposals for the ICD-11 Beta draft:
The deadline in order for proposals to be considered for a frozen version in March/April 2017 was 30 December 2016.
In order for proposals to be considered for inclusion in the version of ICD-11 that is scheduled for release in 2018, they needed to be submitted by March 30, 2017. So those two deadlines have been reached.
Comments by member states and improvements arising as a part of the Quality Assurance mechanism will be included with deadlines later in 2017.
According to Slide #12 in this November 2016 WHO presentation, the deadline for member state comments is May 31, 2017; the deadline for Field Testing and Quality Assurance is June 30, 2017 [2]. But these dates are unconfirmed and may have been revised since November, last year.
Proposals received after the end of May will be considered in the context of ICD-11 maintenance after 2018, when the new version will be subject to an annual update and maintenance schedule [3]. The first annual update is anticipated in 2019.
The Joint Task Force is considering naming each year’s iteration in the format: ICD 2018; ICD 2019; ICD 2020 and so on. There may never be a need for an ICD-12, since an electronic system is better able to evolve “gracefully” – as Dr Christopher Chute (Joint Task Force; Chair, Revision Steering Group) puts it – in response to advances in scientific knowledge and classificatory changes.
Deadlines for submitting comments
I have asked Dr Jakob and the Joint Task Force to clarify by what date comments on proposals that met the March 30 deadline will need to be submitted by in order to be considered in the context of the earliest release of ICD-11, in 2018.
No clarification has been forthcoming; so if you are a stakeholder considering submitting a comment on existing proposals in the Beta draft or on outstanding proposals queued in the “Proposals Mechanism” which are still going through the review process, then I would advise that you put this in hand over the next couple of weeks. If any deadline is announced, I will update at the top of this report.
Frozen release
On April 4, ICD Revision is scheduled to release a frozen version of ICD-11 MMS for field testing*. If there are any changes in this April 2017 Frozen Release that are relevant to stakeholders in the G93.3 terms, I will post an update at the top of this report.
*ICD-11 Field Trials, Information and Terms of Engagement, March 17, 2017 https://hscic.kahootz.com/gf2.ti/af/762498/122441/PDF/-/ICD11_FT_Information_and_ToE.pdf
Current status of the ICD-10 G93.3 legacy categories
The ICD-10 G93.3 legacy categories: Postviral fatigue syndrome; Benign myalgic encephalomyelitis and Chronic fatigue syndrome were taken out of the public version of the Beta draft in early 2013, with no explanation for their absence.
ICD Revision has maintained a cephalopodic grip on its intentions for these terms.
Advocates and patient organization stakeholders have been attempting to obtain transparency from ICD Revision around the Topic Advisory Group for Neurology’s proposals for these terms for over four years. During this period, stakeholders have been disenfranchised from participation in the revision process.
Questions raised in the English Parliament
15 international stakeholder organizations wrote to the ICD-11 MMS Joint Task Force, in February, in support of my call that the Joint Task Force place the matter of the continued absence of proposals for these terms on the Agenda of their February 20–22 meeting, in Cologne.
There were asked to expedite the release of proposals for public scrutiny and comment before the March 30 deadline.
This initiative was met with a disturbing level of obfuscation on the part of WHO and the Joint Task Force, especially given that ICD Revision has been promoted as an open, transparent process, inclusive of stakeholder participation.
The Countess of Mar, a long standing advocate for patients with ME and CFS, tabled two Written Questions in the House of Lords. The first is here (February 27), which received a response that raised more questions than it answered and a follow up question, here (March 16), which received an equally opaque reply.
But on March 26, the three terms were finally restored to the Beta draft – but with this caveat:
“While the optimal place in the classification is still being identified, the entity has been put back to its original place in ICD.”
Team WHO 2017-Mar-26 – 12:46 UTC
This suggests that we should view the restoration of the terms as a “placeholder” and that the work group may release revised proposals later this year.
What do we know?
WHO has confirmed that there is no intention to classify the ICD-10 G93.3 legacy terms under the Mental or behavioural disorders chapter or under the Symptoms, signs chapter.
“Team WHO” has also approved some long standing proposals for exclusions for two of these terms under Fatigue (but not yet approved an exclusion for Postviral fatigue syndrome and I have asked “Team WHO” for the rationale for this apparent anomaly, since one would anticipate that if the inclusion terms are excluded under Fatigue, the ICD concept title entity would also be excluded). Possibly, TAG Neurology has other plans for the classification of PVFS in ICD-11.
So, nearly 10 years into the revision process, it’s still unclear what the work group might be considering for these terms, when they will reach consensus, or whether alternative proposals might be released on April 4, when a frozen version of ICD-11 is scheduled for release for field testing.
How do the terms currently stand in ICD-10?
This is how the G93.3 legacy terms were represented in ICD-10:
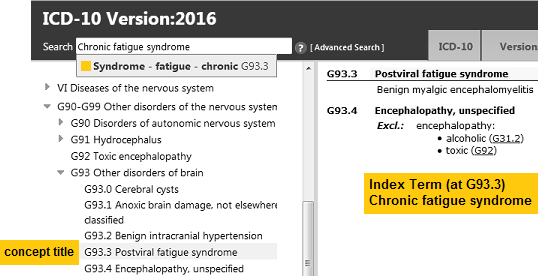
For ICD-10, Postviral fatigue syndrome (PVFS) is the lead (or concept title) term. Benign myalgic encephalomyelitis is the inclusion term under PVFS and takes the G93.3 code. Chronic fatigue syndrome is listed only in the Index, and coded to G93.3.
How do the terms stand in ICD-11 Beta draft, now they have been restored?

Since March 26, 2017, for ICD-11 Beta draft, all three terms are currently back under the Neurology chapter, under parent: Other disorders of the nervous system. PVFS is the lead (or concept title) term. BME and CFS are both specified as inclusion terms to PVFS, in the ICD-11 equivalent of the Tabular List. The terms listed under synonyms and all other “Content Model” descriptive content appear much as the Beta had stood in 2009.
But given the caveat, it is still unknown what the work group might be considering for these terms or whether or when they might release further proposals.
Note that the recommendations of the various external work groups are advisory only. WHO classification experts and the Joint Task Joint can, and sometimes do, overrule work group decisions.
If the Topic Advisory Group for Neurology, that has responsibility for these terms, were to reach consensus and release an alternative set of proposals before 2018, these will not necessarily obtain the approval of WHO/Joint Task Joint.
Suzy Chapman and Mary Dimmock have submitted a proposal
To address this situation, U.S. advocate, Mary Dimmock, and I have collaborated on the preparation of a formal and fully referenced proposal which we submitted on March 27. Our proposal (in the PDF below) recommends that these terms should be retained in the neurological chapter, using separate codes for ME and CFS, and also makes other recommendations.
Our Proposal and Rationale is set out in Part Two:
A proposal for the ICD-10 G93.3 legacy terms for ICD-11: Part Two
For a good overview of ICD-11’s structure and functionality by NHS Digital click here
References:
1 International Statistical Classification of Diseases and Related Health Problems, 11th Revision, Canada, Canadian Institute for Health Information (CIHI). https://www.cihi.ca/en/submit-data-and-view-standards/codes-and-classifications/icd-11
2 Presentation, Dr Robert Jakob, WHO/ICD Revision, November 2016. https://t.co/VvtZXVHZoF
3 ICD Revision Quarterly Newsletter, ICD-11 Update: January 2017. http://www.who.int/entity/classifications/ICD11January2017Newsletter.pdf
